
A volumetric flask is one of the most important pieces of equipment in any analytical or research lab. It looks simple — a flat-bottomed, pear-shaped glass vessel with a long, narrow neck — but that design is deliberate. Every dimension is engineered to serve one specific purpose: preparing a solution to an exact, known volume.
When accuracy matters, a volumetric flask is not interchangeable with a beaker or an Erlenmeyer flask. It is the go-to tool for solution preparation, and understanding how it works makes a real difference in the quality of your results.
Design and Structure
The shape of a volumetric flask is not accidental. The wide, round base holds the bulk of the solution, while the long, narrow neck serves a metrological function — a small change in liquid height in the neck represents a very small change in volume. This tight geometry is what makes precise calibration possible.
A single graduation mark is etched or printed around the neck. This mark indicates the flask’s nominal volume, such as 10 mL, 25 mL, 100 mL, 250 mL, 500 mL, or 1000 mL. Unlike graduated cylinders, which carry multiple markings for approximate measurements, the volumetric flask carries just one — because it is calibrated TC (To Contain) one precise volume, not to measure a range.
For high-precision standards preparation, please refer to our [ Guide to Volumetric Accuracy: Mastering the Meniscus]
Osmosis Scientific volumetric flasks are manufactured in compliance with ISO 1042 and ASTM E288, the primary international standards governing the design, dimensions, and performance tolerances of single-mark volumetric flasks. Flasks come with a ground-glass or plastic stopper for mixing by inversion and to prevent evaporation or contamination during use.
Material: Why Borosilicate Glass Matters
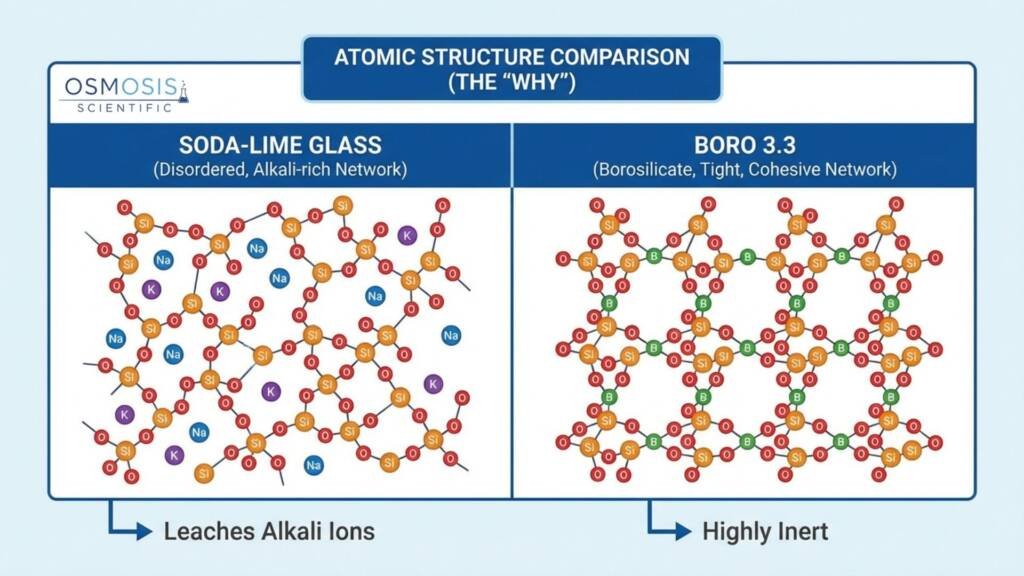
The best volumetric flasks are made from borosilicate glass — and this is not a marketing claim; it reflects a genuine performance difference. Borosilicate glass has a very low coefficient of thermal expansion, which means the internal volume stays dimensionally stable across temperature variations. Soda-lime glass expands and contracts more with temperature, which can introduce measurable errors in precision work.
According to ISO 4787, borosilicate volumetric flasks typically hold their calibration for up to ten years under normal lab conditions, while soda-lime equivalents may require recalibration every five years due to greater susceptibility to chemical surface erosion.
Class A vs. Class B
Volumetric flasks are available in two accuracy grades. Choosing the right one depends on the tolerance your method demands.
| Feature | Class A | Class B |
|---|---|---|
| Tolerance (100 mL) | ±0.10 mL | ±0.20 mL |
| Marking | Permanent, fire-polished | Permanent |
| Calibration standard | ISO 1042 / ASTM E288 | ISO 1042 / ASTM E288 |
| Traceability | Available with Individual Work Certificate (ISO 17025) | Batch certificate |
| Best for | QC labs, pharma, standards prep, titrations | Teaching labs, routine dilutions |
| Typical material | Borosilicate glass | Borosilicate or soda-lime |
Class A has the tightest tolerances and is required for quantitative analysis, reference standard preparation, and calibration workflows. Osmosis Scientific Class A flasks can be supplied with an Individual Work Certificate for ISO 17025 traceability — a requirement in accredited QC and pharmaceutical labs where each flask must have a traceable, documented accuracy record.
Class B has approximately double the tolerance of Class A and suits general teaching labs and less critical bench work where a small added uncertainty is acceptable.
How to Use a Volumetric Flask Correctly
Getting accurate results depends on technique, not just equipment. A few steps make the difference between a reliable measurement and one that carries avoidable error.
Mix thoroughly. After stoppering, invert the flask several times to ensure a homogeneous solution before use.
Dissolve first, then transfer. Dissolve your solute in a small volume of solvent in a separate beaker, then transfer into the flask. Avoid dissolving directly in the flask — exothermic reactions change the liquid volume.
Allow thermal equilibration. Let the solution reach room temperature before making up to the mark. Most volumetric flasks are calibrated at 20°C. A warm solution occupies more volume, so filling to the mark while the solution is still warm produces a more concentrated result than intended.
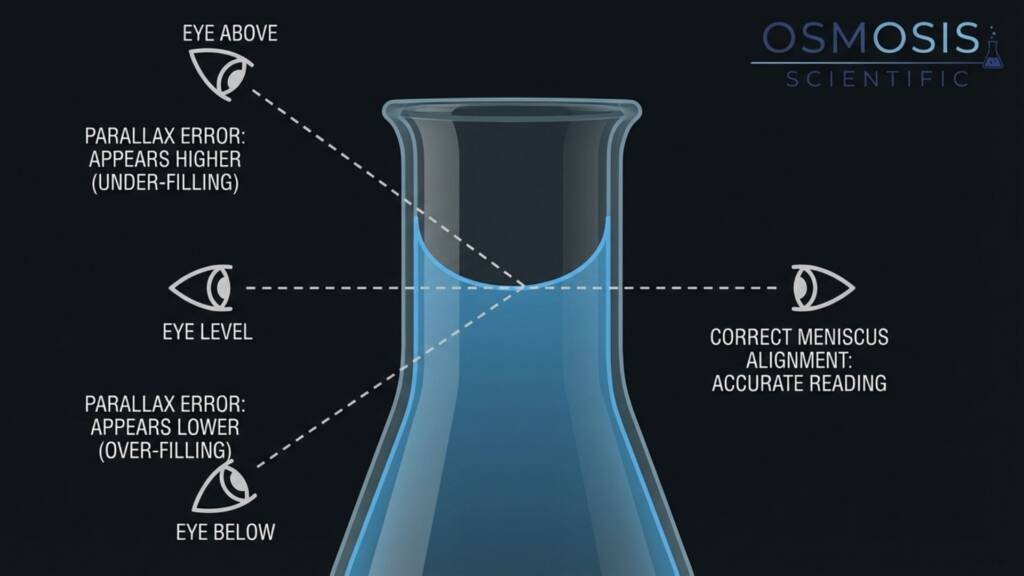
Read the meniscus correctly — and eliminate parallax error. Bring your eye level to the graduation mark and align the bottom of the concave meniscus with the top of the etched line. Aligning the bottom of the meniscus with the graduation mark eliminates parallax error — a common source of volumetric uncertainty where viewing from above or below shifts the apparent position of the liquid surface and introduces a consistent bias into every reading.
Make up to volume carefully. Use a dropper or Pasteur pipette for the final additions when approaching the mark. Adding solvent in bulk increases the risk of overshooting.
Where Volumetric Flasks Are Used
Volumetric flasks appear in nearly every field of quantitative lab work.
In pharmaceutical R&D and quality control, they prepare reference standards and assay solutions to tight tolerances. In clinical and environmental labs, they support calibration standards for regulated test methods. In academic and industrial chemistry, they are the foundation of titration experiments, method development, and solution-based assays where a known concentration is the starting point for everything else.
Because the flask’s accuracy depends entirely on correct use — proper temperature control, careful meniscus reading, and clean glassware — these habits are worth building from the start, whether you’re setting up a new lab or training new staff.
Osmosis Scientific supplies ISO 1042-compliant volumetric flasks across standard capacities and both accuracy grades to meet the needs of research, pharmaceutical, educational, and industrial labs.
Quality Assurance & Liability Notice Products supplied by Osmosis Scientific are produced using high-quality Borosilicate 3.3 glass to ensure superior thermal shock resistance and chemical durability. Volumetric glassware is designed to comply with applicable metrology standards such as ISO 1042 or ASTM E288 where specified.
- Certification: For specialized, certified, or calibration-dependent products, Osmosis Scientific may act as a supplier and distributor for accredited third-party manufacturers. In such cases, certification validity and calibration accuracy remain the responsibility of the original issuing body.
- Proper Handling: Osmosis Scientific shall not be held liable for calibration deviations caused by improper handling, misuse, cleaning errors, or chemical exposure (such as concentrated alkalis) occurring after delivery.
- Sterilization: While Borosilicate 3.3 is autoclavable, users must ensure stoppers are removed or loosened during the cycle to prevent pressure-related damage.
Recommended products
FAQs
A graduated cylinder is designed for versatility, measuring a range of volumes with moderate accuracy. In contrast, a volumetric flask is a single-purpose instrument calibrated to contain one specific volume with high precision. For preparing analytical standards where even a 0.1% error matters, a volumetric flask is the only correct choice.
TC stands for “To Contain.” This means the flask is calibrated to hold exactly the stated volume within its body. Because a small film of liquid (the “wetting residue”) always remains on the glass after pouring, a volumetric flask will always deliver slightly less than its marked volume. For dispensing exact volumes, a pipette or burette (calibrated TD – To Deliver) should be used instead.
Precision glassware is calibrated at a reference temperature, usually 20°C. Because liquids expand when heated, a warm solution will appear to hit the graduation mark but will actually contain less mass than intended once it cools. To maintain the integrity of your molarity or concentration, always allow solutions to reach room temperature before the final “make-up” to the mark.
Direct heating (like a Bunsen burner) is strictly prohibited, as it can cause permanent dimensional changes to the glass, ruining the calibration. While Borosilicate 3.3 flasks are autoclavable for sterilization, you must remove or loosen the stopper. A sealed flask creates a pressure differential during the cooling phase that can lead to glass fatigue or implosion.
Clean using a non-abrasive detergent and rinse thoroughly with deionized water. Never use wire brushes, as micro-scratches on the neck can distort the meniscus and alter the glass’s wetting properties. Store flasks upright and uncapped; trapping moisture inside can lead to “alkaline leaching” over time, which may etch the glass and eventually shift the calibrated volume.






