A burette is one of those classic lab instruments that quietly does the hard work of precision. It looks simple—a long, narrow tube with a tap at the bottom—but it sits at the heart of accurate titrations and quantitative analysis in chemistry labs around the world. From the Osmosis Scientific point of view, this Laboratory Burette Guide will help you understand how a burette works, where it’s used, and what its main components are, ensuring you choose the right model for your lab.
What is a burette?
In basic terms, a burette (or buret) is a long, straight, graduated tube with a stopcock at the bottom and a fine tip that allows you to deliver a liquid in a controlled way—either as a steady stream or drop by drop. It is classified as volumetric glassware because its primary purpose is to measure and dispense very precise volumes of liquid (and in some designs, gas) rather than just to hold solutions.
The graduation scale usually runs from 0 mL at the top down to the full capacity (for example, 25 mL, 50 mL or 100 mL) at the bottom, with fine subdivisions such as 0.1 mL or better. During use, you read the liquid level at the meniscus, deliver the titrant through the stopcock and tip, and calculate the volume dispensed from the difference between the initial and final readings. Burettes are calibrated “To Deliver” (TD), meaning the indicated volume corresponds to what actually leaves the tip when the instrument is used correctly.
Osmosis-style takeaway: if your lab work depends on small differences in concentration, a burette is the tool that lets you see those differences clearly instead of guessing from bulk measurements.
If you want to learn How to Read a Burette Correctly
Main components of a burette
Even the simplest manual burette is a carefully engineered system. The key components you’ll handle every day are:
1. Graduated tube (barrel)
The main body is a long, uniform cylindrical tube made of glass or clear plastic, with volume graduations etched or printed along its length.
- The scale is usually arranged with 0 mL at the top and the maximum volume at the bottom, which suits titration readings where you subtract initial from final volume.
- The internal bore of the tube must be extremely uniform; any taper or irregularity would make the delivered volume per unit length non‑linear and break the calibration.
High‑quality burettes are commonly made from borosilicate glass because it offers excellent chemical resistance, low thermal expansion and the ability to withstand autoclaving without losing calibration. For student or field work, plastic versions (acrylic or PMP) offer better break resistance at the cost of slightly lower accuracy.

Some advanced glass burettes include a Schellbach stripe: a vertical white band with a narrow blue line on the back of the tube that makes the meniscus much easier to read accurately.
2. Stopcock (valve)
The stopcock is the control centre for flow. It sits near the bottom of the tube and lets you open, close or partly open the passage to regulate how fast the liquid leaves.
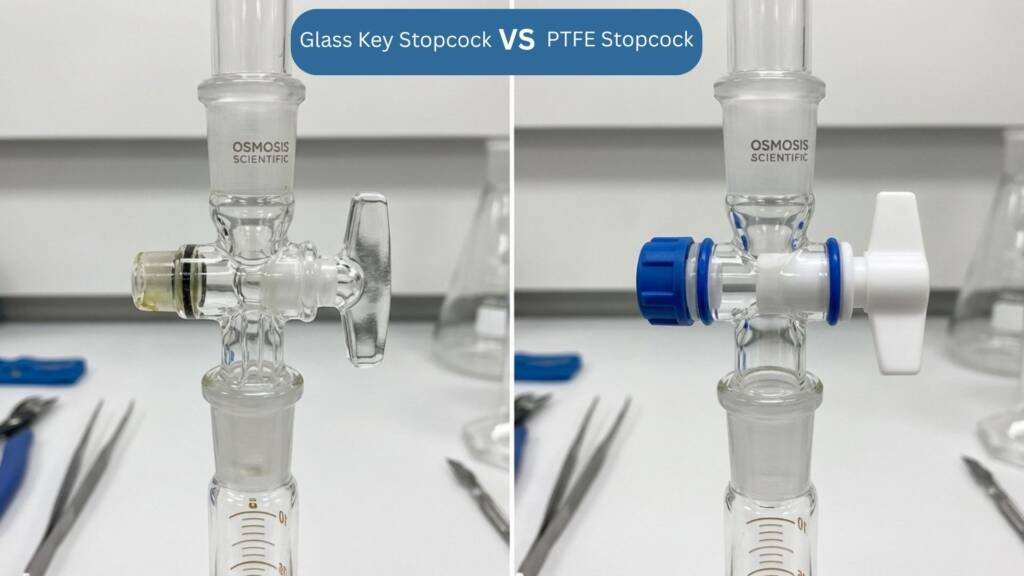
Two main designs are common:
- Glass key stopcocks use a ground‑glass plug in a matching barrel. They give a good seal but need a thin layer of grease to rotate smoothly and stay leak‑free. Over time, that grease can harden or migrate, so they require regular cleaning and re‑lubrication.
- PTFE (Teflon) stopcocks are now widely used. PTFE is self‑lubricating and chemically inert, so these stopcocks don’t need grease and are easier to maintain while also reducing contamination risk. Some systems use a screw‑type “Rotaflow” needle valve for extremely fine flow control.
Getting this part right matters: a poor stopcock will leak, jerk or clog, and all of those show up as inconsistent titration results.
3. Capillary tip
Below the stopcock is the narrow, tapered tip. Its job is to turn the stored volume into manageable drops.
- The constriction creates back pressure so small movements of the stopcock correspond to gentle changes in flow.
- A fine tip produces small, regular droplets, which is vital near the titration endpoint where a single drop can change the indicator colour.
For manual work, the smallest practical addition is usually one drop or even a controlled half‑drop touched onto the side of the receiving flask.
4. Graduations and markings
Good burettes rely on clear, precise markings:
- Graduations often every 0.1 mL, allowing you to estimate volumes to the nearest 0.01–0.02 mL in careful work.
- Many burettes are manufactured to Class A or Class B standards, with Class A offering tighter tolerances for analytical applications and Class B intended for routine or teaching work.
In regulated labs, you’ll also see serialization and calibration certificates, which matter for audits and method validation.
5. Top opening and accessories
The open top allows filling with titrant, usually using a small funnel to avoid spills. A burette is typically used with:
- A stand and burette clamp to hold it strictly vertical during operation.
- In some teaching or industrial setups, an integrated frame or protective casing for extra stability.
Digital and electronic burettes add extra parts—pistons, drive mechanisms, displays and recirculation valves—but conceptually they still combine a reservoir, a flow‑control system and an outlet tip.
Practical uses of a burette
From the Osmosis Scientific perspective, burettes are most important wherever your lab needs accurate, repeatable dosing of a liquid reagent. In this section of our Laboratory Burette Guide, we group the uses naturally into a few big buckets.
1. Classical titration experiments
This is the textbook application and still the main one in modern labs. The burette holds a standard solution (titrant) of known concentration, which you add to your sample (analyte) until the reaction endpoint is reached, often signalled by an indicator colour change.
Common titration types include:
- Acid–base titrations – for determining the molarity of acids or bases in samples like HCl, NaOH or buffer systems.
- Redox titrations – for oxidizing or reducing agents such as permanganate or dichromate in environmental or industrial samples.
- Complexometric titrations – using ligands like EDTA to measure metal ions, for example calcium and magnesium in water hardness tests.
- Precipitation titrations – where the endpoint is defined by the formation or disappearance of a precipitate, such as halide determinations.
Here, the burette’s precise graduations and smooth flow control make the difference between “roughly right” and truly quantitative.
2. General quantitative chemical work
Beyond classic titration, burettes are used whenever you need to add a known, adjustable volume of reagent very precisely. Typical scenarios include:
- Fine volume adjustments to reach a specific stoichiometric point or maintain a constant pH in pH‑stat titrations.
- Calibrating other volumetric apparatus by delivering test volumes and checking their readings.
- Preparing exact standard solutions by carefully dosing concentrated reagents into diluents.
In short, if your protocol says “add until…”, there’s a good chance a burette is the safest way to do it accurately.
3. Industrial and applied laboratory uses
Because titration underpins so much quality control, burettes turn up in many industries:
- Pharmaceuticals – assay and purity tests for active pharmaceutical ingredients, content uniformity checks across tablet batches, and back‑titration in cases where the drug is poorly soluble or reacts slowly.
- Food and beverage – acidity measurements in wines, juices and dairy products, vinegar analysis and control of preservatives and additives.
- Cosmetics – determining concentrations of surfactants, oils and actives to ensure each batch meets specification.
- Automotive and chemicals – analysing lubricants, fuels, coolants and plating baths to maintain correct formulation and detect degradation.
- Environmental and water testing – measuring water hardness, alkalinity, chloride, dissolved oxygen (e.g. Winkler titration) and other routine water‑quality parameters.
- Education and research – teaching the fundamentals of volumetric analysis as well as performing precise reaction studies and method development.
If your lab runs regulated QC or accreditation‑driven tests, burette‑based titration is often still written directly into the standard methods.
Types of burettes you might encounter
While the classic glass burette is the default, your choice of type affects accuracy, durability and workflow. Osmosis‑style selection usually considers the following categories.
1. Manual volumetric burettes
These are the familiar gravity‑fed instruments in most labs:
- Macro burettes (25–100 mL) for general titrations in the 10–40 mL range.
- Micro burettes (1–10 mL) with very fine subdivisions for small sample volumes and high‑precision work.
- Automatic self‑zeroing burettes attached to a reservoir, which fill via a pump or bulb and reset the meniscus to zero automatically to improve throughput and reduce setup errors.
2. Digital and electronic burettes
Piston‑driven burettes combine a traditional titration setup with digital measurement:
- A piston moves inside a cylinder to dispense liquid, while an encoder tracks the volume and shows it on an LCD.
- Features like recirculation valves allow priming without wasting expensive reagents and USB/RS232 connectivity supports automated data logging in regulated environments.
These are especially useful in pharmaceutical and QC labs where traceability and consistency are critical.
3. Gas burettes and specialised designs
Some burettes are adapted for measuring gases or supporting specific industrial analyses:
- Gas burettes (e.g. Hempel designs) measure gas volume by displacement of a confining liquid through a connected leveling tube.
- Tutwiler gas burettes are specialised tools for determining hydrogen sulfide content in fuel gases using iodimetric titration, crucial in petrochemical safety and compliance.
In all of these variations, the underlying principle remains the same: controlled, measurable delivery of a known reagent.
Summary: Why this Laboratory Burette Guide Matters
Even as automated titrators and fully digital systems become more common, the burette keeps its place as a practical, flexible and surprisingly affordable backbone for volumetric analysis. We hope this Laboratory Burette Guide has demonstrated that high-quality borosilicate burettes are critical for credibility in your data.
Ready to upgrade your lab’s precision? Explore our full range of Class A Borosilicate Burettes at Osmosis Scientific.Request a Bulk Quote for Exports
What is a burette used for in the laboratory?A burette is mainly used for titration experiments and other quantitative analyses where you must deliver an accurately measured, adjustable volume of liquid reagent into a sample.
Why is a burette preferred over a measuring cylinder or pipette?Unlike a cylinder, a burette offers fine graduations and precise flow control, and unlike a volumetric pipette it can deliver any volume up to its capacity while still being measured accurately.
What are the main components of a burette?The key components are the graduated tube (barrel), stopcock valve, narrow capillary tip, volume graduations/markings, and the top opening often used with a funnel and stand.
What is the difference between Class A and Class B burettes?Class A burettes are manufactured with tighter tolerances and are intended for high‑precision analytical work, while Class B burettes have wider tolerances and are suited to teaching or routine tasks.
Can burettes be used outside academic labs?Yes—burettes are standard tools in pharmaceutical QC, food and beverage testing, cosmetics, automotive and chemical industries, as well as environmental and water‑quality laboratories.


