In physical metrology and laboratory practice, the meniscus refers to the curved surface of a liquid within a container. While it may appear to be a simple visual artifact, the meniscus is the fundamental interface for volumetric measurement. Its formation is governed by the physical laws of surface tension and capillary action, and its accurate interpretation is critical for experimental validity. This guide outlines the importance of the meniscus in laboratory glassware, detailing the mechanisms of precision, the prevention of error, and the best practices for ensuring reproducibility in scientific analysis.
1. The Physics of the Meniscus
To understand the importance of the meniscus, one must first define its physical origin. The meniscus is the curve formed at the upper surface of a liquid in a container, resulting from the interplay of two intermolecular forces:
- Cohesion: The attractive force between like molecules within the liquid.
- Adhesion: The attractive force between the liquid molecules and the container walls.
Capillary Action
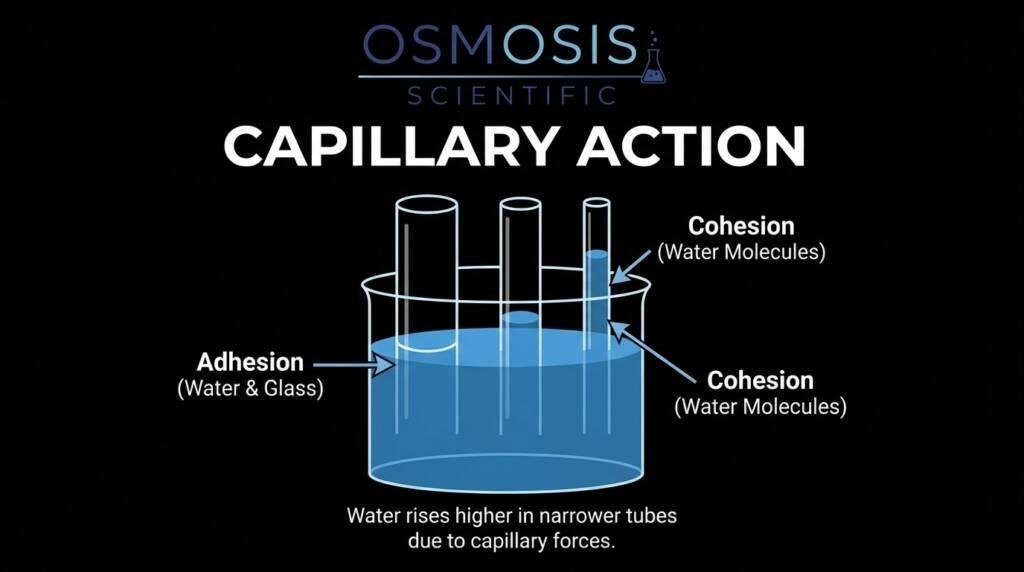
Capillary action causes the liquid to rise slightly along the vertical walls of the glassware. This phenomenon is particularly strong in liquids that “wet” the glass, such as water or aqueous solutions.1 This interaction creates the characteristic curve—usually concave (curved downward) for water in glass—which serves as the reference line for all volumetric measurements. Understanding this behavior is essential for selecting the correct measurement protocols for liquids that interact differently with container surfaces.
2. Key Pillars of Importance
According to technical literature from laboratory glassware manufacturers, the importance of the meniscus rests on five main pillars: precision, consistency, error avoidance, capillary mechanics, and environmental compensation.
2.1 Precision in Volume Measurement
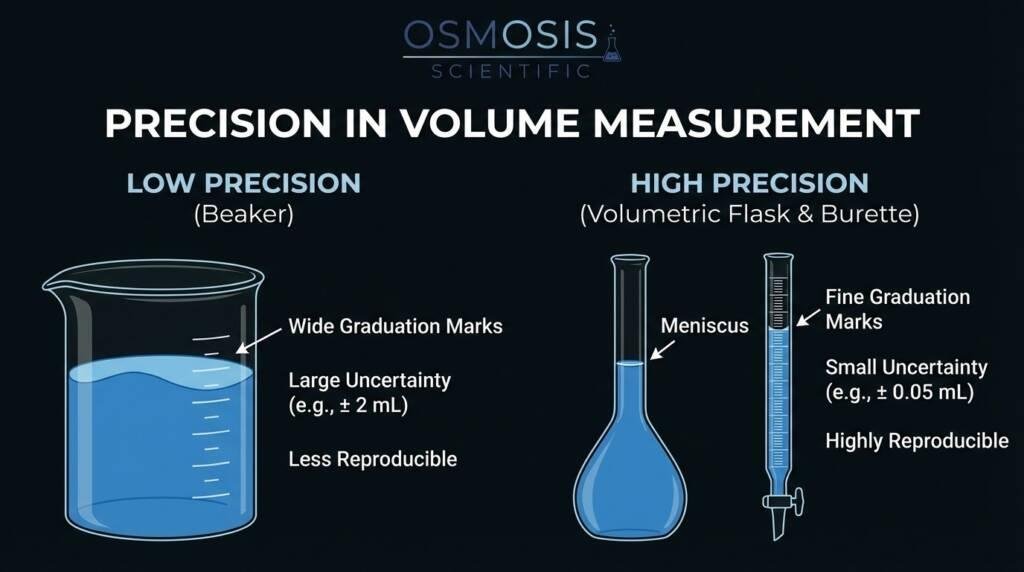
The primary function of the meniscus is to act as the definitive indicator of volume. In high-precision Osmosis Scientific glassware—such as volumetric flasks, burettes, and graduated cylinders—the calibration marks are designed to align with a specific part of the liquid curve.
- The Rule of the Nadir: For the vast majority of laboratory liquids (which form a concave meniscus), precision is achieved only by reading the volume at the bottom (or nadir) of the curve.
- Accuracy: Reading the lowest point ensures that the volume recorded matches the calibrated capacity of the vessel. Deviating from this point introduces a systematic error that compromises the accuracy of the entire experiment.
2.2 Avoiding Parallax Errors

One of the most common sources of experimental error is parallax—the apparent shift in the position of an object when viewed from different angles.
- The Mechanism of Error: If a technician looks down at the meniscus, it will appear higher than the graduation mark; if they look up, it will appear lower.
- The Solution: The distinct line of the meniscus bottom serves as a visual anchor. By positioning the eye exactly level with the bottom of the meniscus, the observer eliminates angular distortion. This practice is non-negotiable for obtaining accurate readings.
2.3 Consistency and Reproducibility
Science relies on reproducibility. If one researcher reads the top of the curve and another reads the bottom, their data will not be comparable.
- Standardization: By adhering to the universal standard of reading the bottom of the meniscus (for concave liquids), scientists ensure consistency across different experiments and laboratories. This consistency is crucial for the reliability of analytical results.
2.4 Compensation for Temperature Variations

Liquid volumes are sensitive to temperature changes. The meniscus plays a role in accounting for these fluctuations.
- Thermal Expansion: Changes in temperature cause liquids to expand or contract. Reading the volume at the bottom of the meniscus helps to account for these thermal variances, ensuring that the measurement remains robust despite minor environmental shifts.
3. Best Practices for Reading the Meniscus
To respect the importance of the meniscus and ensure data integrity, the following protocols should be observed:
- Eye Level Alignment: Always position the eye at the exact level of the liquid surface.
- Contrast Enhancement: For clear liquids, place a white card or a “meniscus reader” (a card with a black stripe) behind the glassware, just below the fluid level. This renders the profile of the meniscus dark and sharp against the light background.
- Vertical Stability: Ensure the glassware (burette or cylinder) is perfectly vertical. Tilted glassware distorts the meniscus shape and the corresponding volume reading.
Cleanliness: A distorted or irregular meniscus often indicates dirty glassware. Grease or chemical residues interfere with adhesive forces, preventing the smooth formation of the curve. Glassware must be chemically clean to ensure the meniscus forms correctly for an accurate reading.
In the laboratory, the meniscus is more than a curve; it is a calibration tool. Its importance lies in its ability to standardize the interface between a liquid and a measurement scale. By strictly adhering to the protocols of reading the meniscus—specifically utilizing the bottom of the curve to avoid parallax and ensure precision—researchers safeguard the validity of their volumetric analyses. At Osmosis Scientific, we emphasize that paying attention to this small detail is a critical practice that underpins the reliability of all scientific inquiry.
| Measurement Factor | Best Practice | Scientific Reasoning |
| Reading Point | Bottom of the curve (Nadir) | Aligns with calibration index |
| Eye Level | Exactly level with liquid surface | Eliminates parallax distortion |
| Temperature | Maintain 20°C (standard) | Minimizes thermal expansion error |
| Cleanliness | Chemically clean glass | Ensures smooth meniscus formation |
FAQ’s
For concave liquids like water or aqueous solutions, the lowest point of the curve—the nadir—is the only part of the liquid that aligns with the calibrated graduation marks of high-precision glassware. Reading from any other point introduces a systematic error that compromises the accuracy of your entire experiment.
Parallax error occurs when your eye is not level with the liquid surface, making the volume appear higher or lower than it actually is. To eliminate this angular distortion, always position your eye at the exact level of the bottom of the meniscus
A distorted or irregular meniscus is often a sign of dirty glassware. Grease or chemical residues interfere with the adhesive forces between the liquid and the glass, preventing the smooth formation of the curve and leading to sheeting errors.
Yes, liquid volumes are sensitive to thermal changes. Changes in temperature cause liquids to expand or contract; therefore, reading the volume consistently at the bottom of the meniscus helps account for these variances and ensures robust data despite minor environmental shifts.



1 Comment