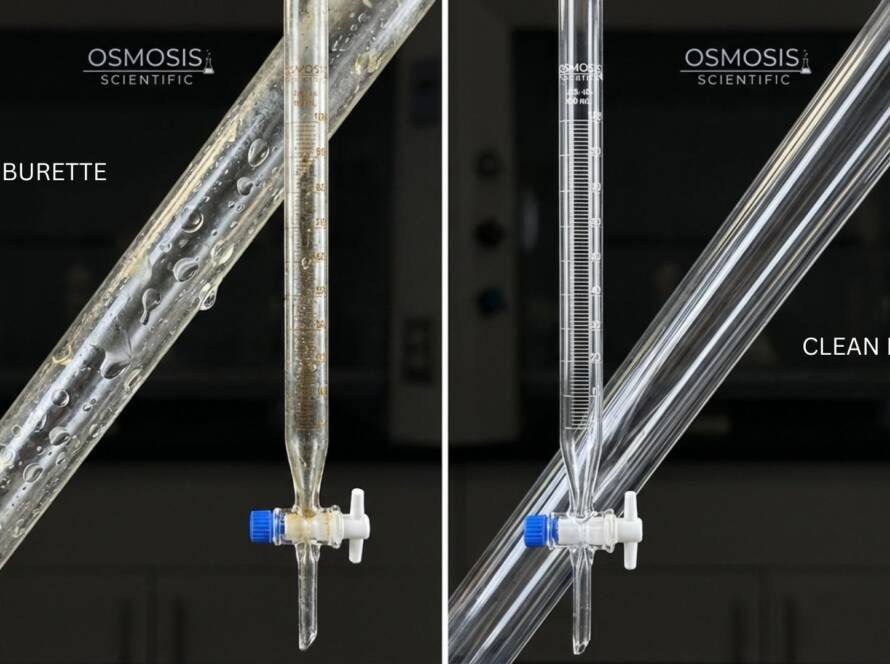
Even “glass” isn’t a singular material in the lab. Borosilicate 3.3 and Soda-Lime behave very differently under heat and chemicals, and that divergence directly determines day-to-day lab safety. The most important safety lever is thermal expansion: Borosilicate 3.3 expands far less with temperature, building significantly less internal stress, making it much less likely to shatter during heating, cooling, or sterilization cycles.
Laboratory accidents rarely happen because someone bought the wrong beaker size. They happen because a vessel experiences thermal gradients (hot base, cooler walls), chemical attack, or cumulative micro-damage—and the glass type cannot tolerate the stress. Selecting Borosilicate 3.3 for heated and chemical workflows is a safety decision as much as it is a purchasing one.
The Atomic Foundation: Network Formers and Stability
The behavioral differences between these glasses originate at the molecular level.
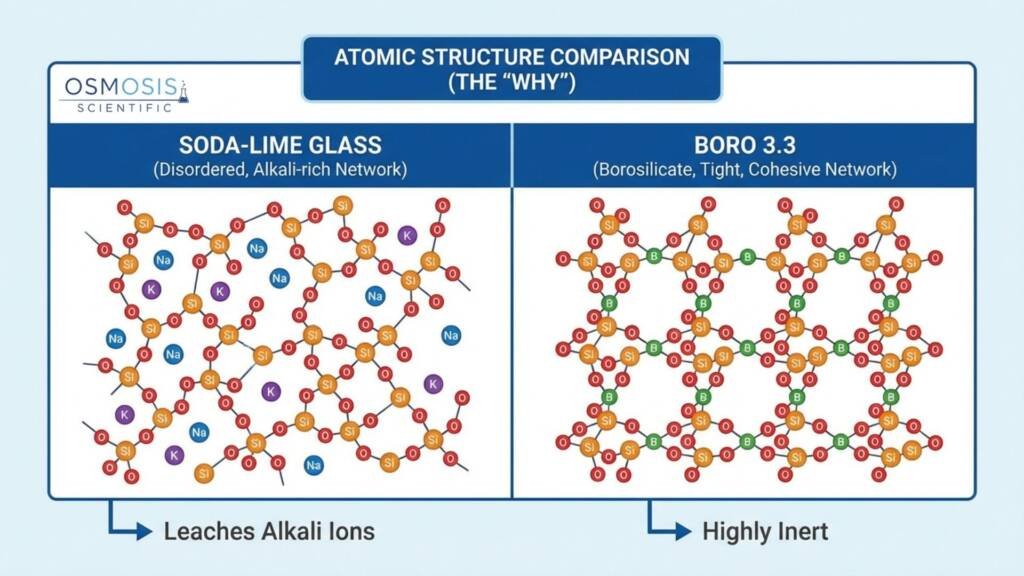
- The Structural Limitations of Soda-Lime: Soda-Lime glass (Type III) uses “modifiers” like sodium oxide (soda) and calcium oxide (lime) to lower its melting point. This disrupts the continuous silica bonds, creating a high concentration of mobile sodium ions that make the glass prone to leaching and expansion.
- The Engineering of Borosilicate 3.3: Borosilicate 3.3 (Type I) strategically replaces most alkali modifiers with boron trioxide (B₂O₃). Boron atoms integrate into the silica network to create a tighter, more robust covalent structure that maintains dimensional stability even under intense stress.
The Hook: Thermal Shock Starts with Expansion
Key Focus: Cubical Expansion and Safety Envelopes
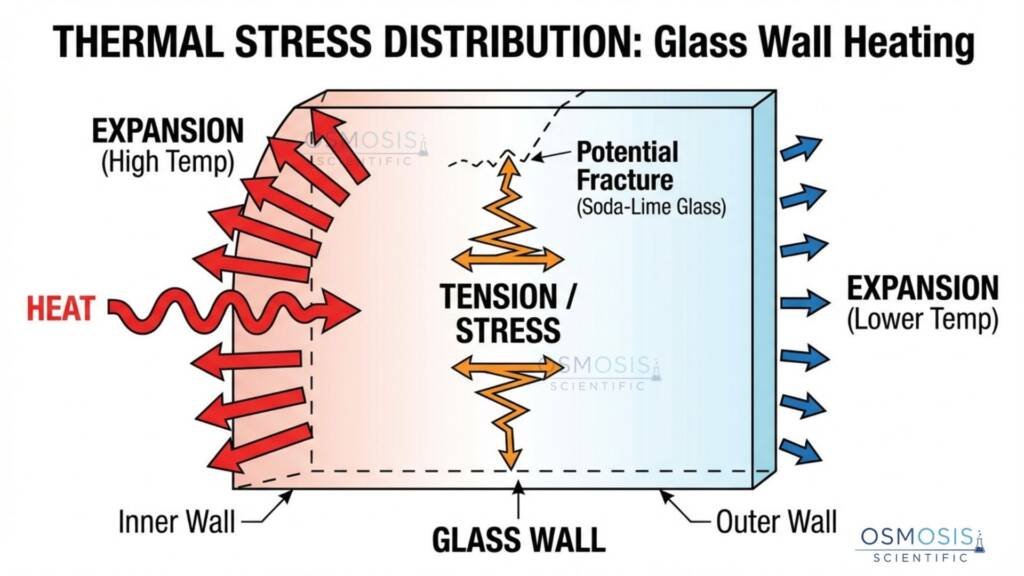
Comparing Coefficient of Cubical Thermal Expansion (γ)—the volumetric expansion—reveals the true safety gap:
- Borosilicate 3.3: ≈ 9.9 × 10⁻⁶ °C⁻¹ cubical expansion.
- Soda-Lime Glass: ≈ 27 × 10⁻⁶ °C⁻¹ cubical expansion.
This means Soda-Lime expands at roughly 2.7× the volumetric rate of Borosilicate for the same temperature rise. In real workflows—not media prep, autoclaving, or quick transitions from storage to bench—lower expansion translates to a larger safety envelope and fewer sudden failures.
Why this matters for Beakers and Erlenmeyer Flasks
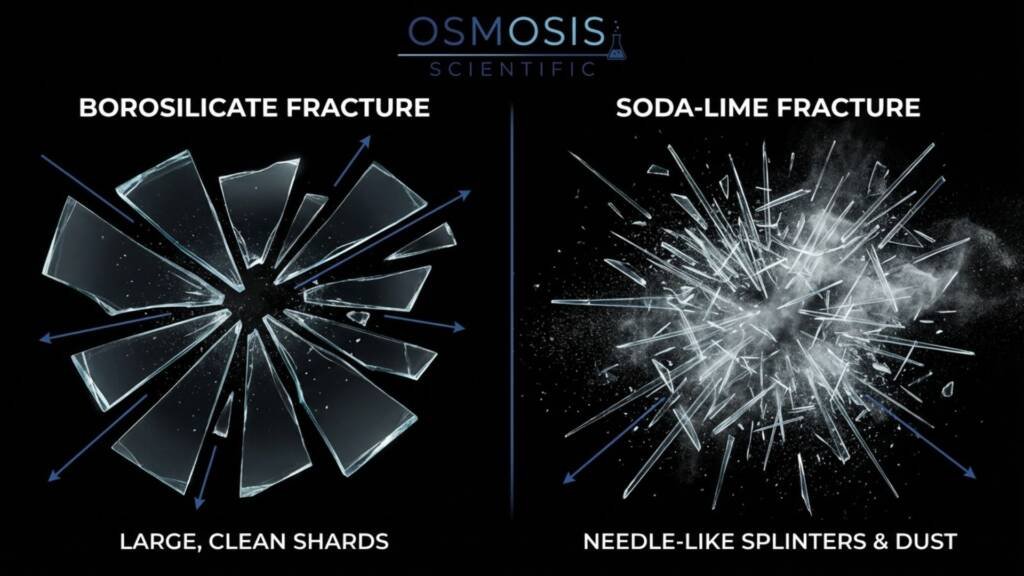
These vessels are routinely exposed to conditions that create thermal gradients: hot plate hot spots and repeated sterilization cycles. Because Soda-Lime builds stress faster under these gradients, it is more prone to unpredictable cracking—especially if it has micro-scratches that act as crack starters. Borosilicate 3.3, with a thermal shock resistance of 100 K (vs. 30 K for Soda-Lime), is the only safe default for general-purpose heated labware.
Chemical Resistance and “Silent” Safety Risks

Safety isn’t just about breakage; it includes what the container contributes to your solution.
- Soda-Lime: Contains higher mobile alkali ions, causing pH fluctuations and contamination that can ruin delicate assays.
- Borosilicate 3.3: Engineered with low alkali content and a robust network, it is classified as USP Type I (Neutral) glass, making leaching negligible.
Practical Buying Checklist (For Lab Managers)
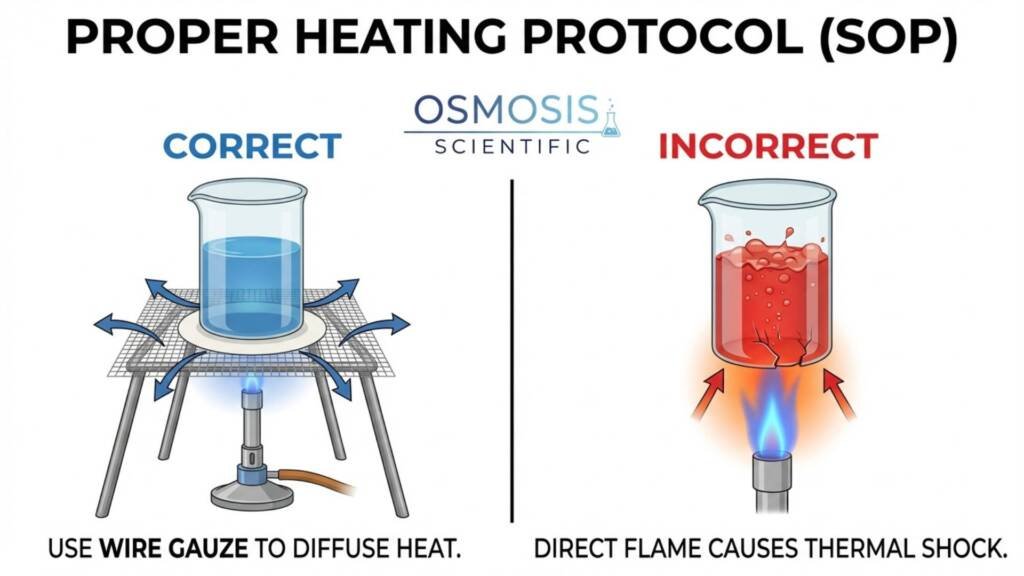
Use this quick decision filter when specifying glassware for your facility:
- Choose Borosilicate 3.3 for: Hot plates, boiling, autoclaves, vacuum filtration, and routine heated sample prep.
- Restrict Soda-Lime to: Low-heat, low-risk, non-critical storage and basic handling at room temperature.
- Inspect and Retire: Scratches, chips, and “frosting” (clouding) multiply thermal-shock risk.
- Avoid Abrasives: Metal-bristle brushes create micro-scratches that lead to future failures under heat.
FAQs
Borosilicate 3.3 has much lower thermal expansion, so it develops less internal stress during heating/cooling and is far less likely to crack from thermal shock. Soda‑lime expands faster and can fracture unpredictably under localized heating or rapid temperature changes.
Cubical (volumetric) thermal expansion tells how fast the glass’s internal volume changes per °C, and higher values amplify stress when temperature is uneven across the vessel. With 9.9 × 10 − 6 ∘ C − 1 9.9×10 −6 ∘ C −1 for borosilicate vs. 27 × 10 − 6 ∘ C − 1 27×10 −6 ∘ C −1 for soda‑lime, soda‑lime builds thermal stress much faster in hotplate/flame scenarios.
Soda‑lime glass has a much lower thermal ceiling (often cited around 120–150 °C) and is considered unsuitable for direct heating because it cannot tolerate localized heat loads safely. For heated work, borosilicate 3.3 is the fit‑for‑purpose choice due to its thermal shock resistance and higher safe operating temperature.
Soda‑lime contains more mobile alkali ions (like sodium and potassium), which can leach into solutions via ion exchange—especially with water and acidic/alkaline media. This can lead to pH shifts and interference in sensitive assays or analytical workflows.