
Accurate volumetric measurements are the foundational integrity of quantitative chemistry.When results drift or fail to match theoretical values, the culprit is often not the chemistry itself, but a handful of repeatable technique and glassware errors. In high-precision analytical work, maintaining an error margin of 0.1% requires a rigorous understanding of the variables influencing liquid handling.
Even the most accurate Class A flask will “lie” to you if it is mistreated. Here are the five most common mistakes that quietly destroy volumetric accuracy—and how to fix them.
1. The Temperature Trap: Thermodynamic Fluctuations
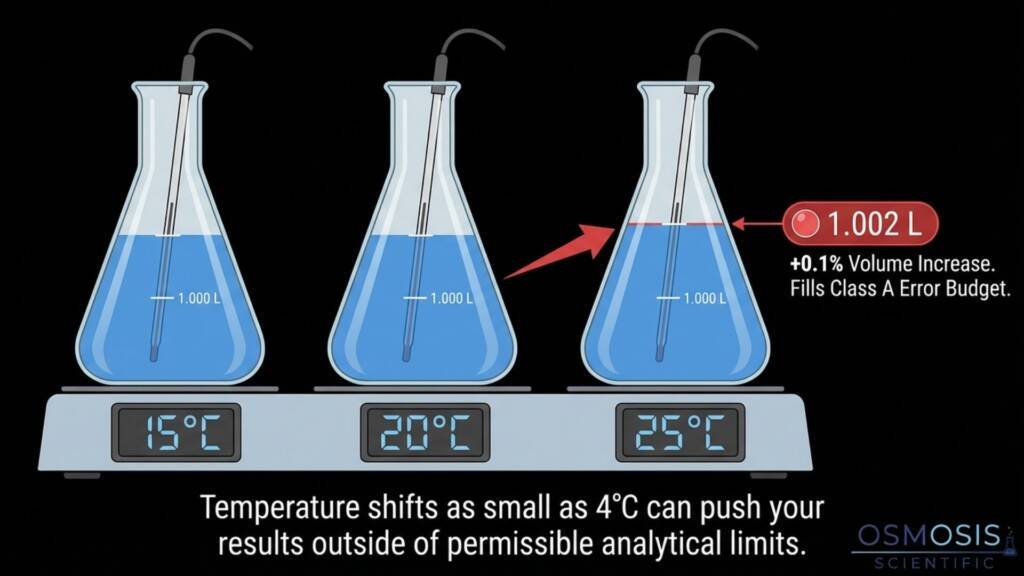
Temperature is the most pervasive source of volumetric error, affecting the liquid, the vessel, and the environment.
Most instruments are calibrated at 20°C (or 25°C to 27°C depending on regional standards).
- The Error: Using solutions that haven’t reached thermal equilibrium.
Dilute aqueous solutions expand by approximately 0.00025°C⁻¹.
This means a 4°C temperature shift is enough to consume a 0.1% error budget. - The Oven Risk: Placing volumetric flasks in hot drying ovens is dangerous.
While borosilicate glass has a low expansion coefficient (9.9 × 10⁻⁶ °C⁻¹), exposure above 150°C can cause permanent calibration drift. - How to Avoid It:
- Allow glassware and reagents to reach thermal equilibrium with the lab for at least one hour before measurement.
- Avoid drying volumetric flasks at high temperatures; if necessary, stay well below 150°C.
- Use density tables or Z-factor corrections if working away from the calibration temperature.
2. The Meniscus Mistake: Parallax and Visual Alignment
The interface between a liquid and its container is governed by surface tension and adhesive forces, forming a meniscus.
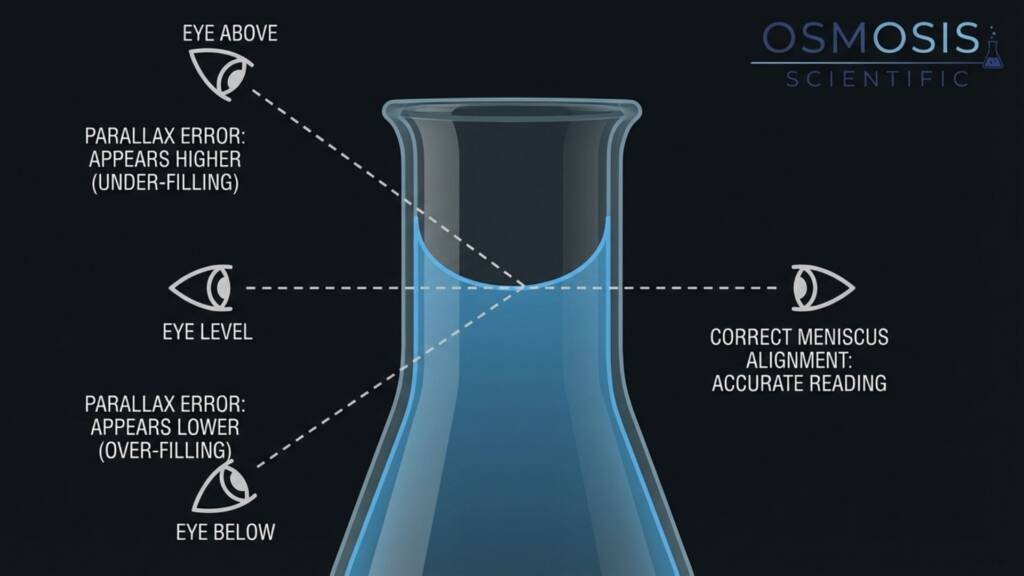
- The Error: Parallax error is an observational distortion that occurs when your eye is not in the same horizontal plane as the graduation mark and the meniscus.
Viewing from an upward angle leads to under-filling, while a downward angle causes over-filling.
In wide-neck vessels, this can easily exceed 1–2% volume error. - How to Avoid It:
- Bring your line of sight perfectly perpendicular to the graduation mark.
- For transparent liquids, align the bottom of the concave meniscus with the top edge of the graduation line.
- Use a meniscus reader—a card with a black and white horizontal split—behind the graduation mark to provide a sharp reflection of the meniscus edge.
3. Dirty, Wet, or Chemically Damaged Glassware
Volumetric glassware is only as accurate as its internal surface.

- The Error: Oily fingerprints or detergent films change the contact angle, preventing proper wetting and distorting the meniscus.
- The “Frosting” Effect: Glass is not entirely inert.
Strong alkalis and hydrofluoric acid etch the interior of glass vessels, a process known as frosting.
This removes wall material and measurably increases internal volume over time.
Soda-lime glass is particularly susceptible, showing changes within 5 years. - How to Avoid It:
- Inspect for “frosting” or droplets before use; clean thoroughly and rinse with deionized water.
- Handle by the body, not the neck, to avoid oily skin contact with calibration areas.
- Remove droplets above the calibration line with a lint-free tissue without wicking below the mark.
4. Misapplication of TC vs. TD and Pipetting Technique
Technicians often fail to distinguish between instruments designed To Contain (TC/In) and those To Deliver (TD/Ex).
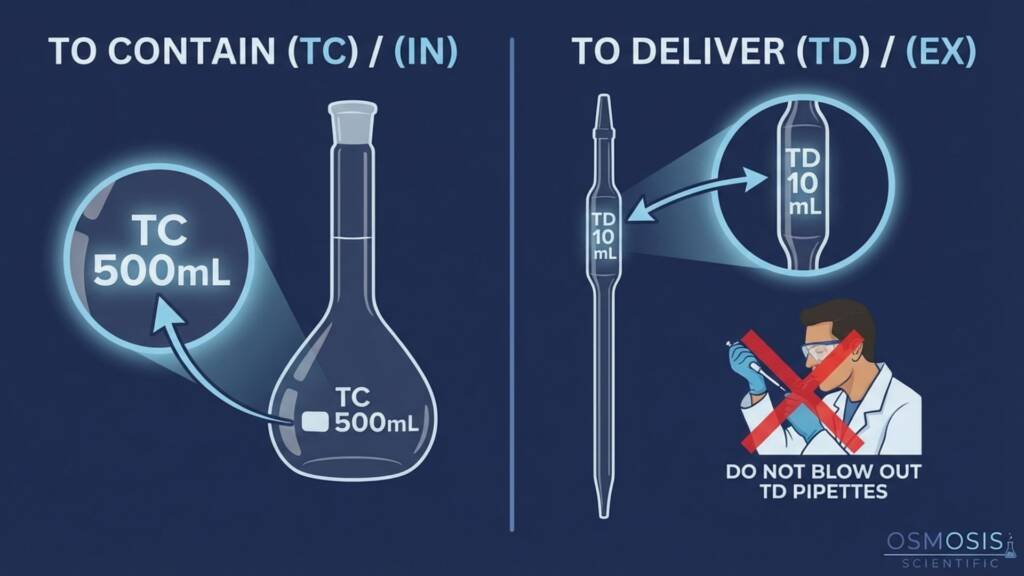
- The Error: Volumetric flasks are TC; they hold the indicated volume but deliver less due to the residual film left on the walls.
Pipettes and burettes are TD; their markings account for this residual drainage film.
A critical mistake is “blowing out” a TD pipette that was designed for gravity drainage. - Piston-Operated Pipettes: Errors also occur through “hand warming,” where heat from an operator’s palm expands the internal air cushion, causing lower aspiration volumes.
- How to Avoid It:
- Use TC vessels for dilutions and TD devices for transfers.
- For viscous liquids like glycerol, use reverse pipetting (depressing the plunger to the second stop before aspiration) to compensate for film loss.
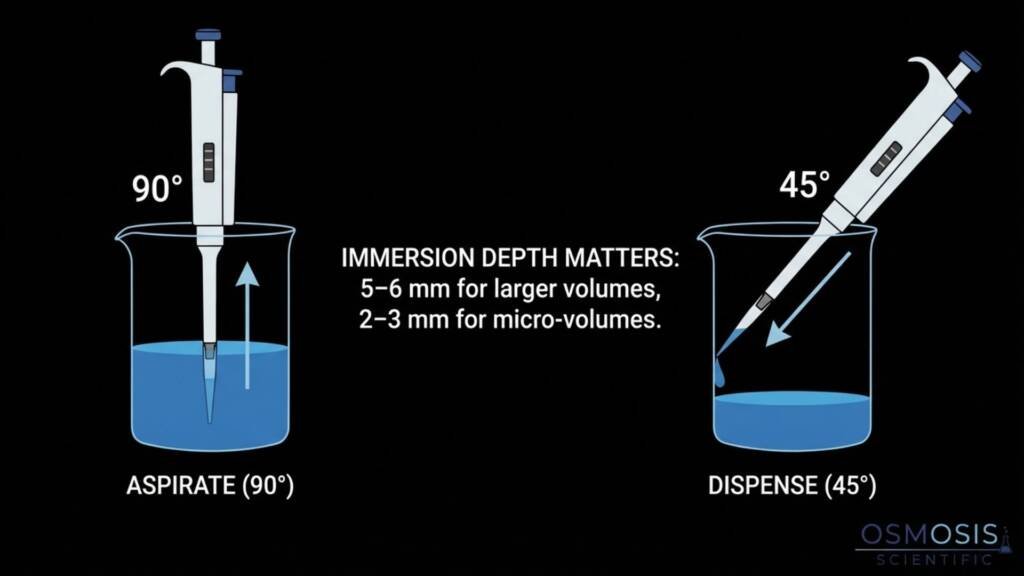
- Hold pipettes vertically (within 20° of 90°) during liquid intake to maintain proper hydrostatic pressure.
5. Skipping Calibration and Maintenance Discipline
Assuming factory calibration is permanent is a major source of systematic error.
- The Error: Mechanical parts in pipettes, such as O-rings and seals, degrade over time, leading to air leaks and significant under-delivery.
Additionally, unstandardized titrants like sodium hydroxide can absorb CO₂ from the air, invalidating calculations. - How to Avoid It:
- Periodically verify performance gravimetrically by weighing dispensed water and applying the buoyancy correction.
- Implement a recalibration schedule every 3 to 12 months.
- Standardize titrants at appropriate intervals rather than trusting nominal label concentrations.
Strategy for Error Elimination
To achieve the 0.1% accuracy goal, the professional laboratory should follow this protocol:
- Environmental Stabilization: Equilibrate all tools and reagents for at least one hour.
- Apparatus Verification: Select the correct TC or TD tool and check for frosting.
- Technique Standardization: Enforce drainage times (15–20 mL/min for burettes) and proper aspiration angles.
- Rheological Adaptation: Use pre-wetting for volatile solvents and reverse pipetting for viscous liquids.
- Metrological Oversight: Apply Z-factor models in gravimetric testing to account for liquid density, air buoyancy, and glass expansion.
FAQs
While borosilicate glass is durable, subjecting it to temperatures above 150°C can cause the glass to drift from its original factory calibration. Even if the flask doesn’t break, this thermal stress can permanently change its internal volume, meaning your “Class A” measurements will no longer meet the required precision standards.
The most frequent mistake is parallax error, which occurs when you eye is not perfectly level with the graduation mark and the liquid’s surface. If viewed from above, the meniscus appears higher (leading to under-filling), and if viewed from below, it appears lower (leading to over-filling). In wide-neck vessels, this visual misalignment can introduce an error as high as 1–2%.
Technically, no. A volumetric flask marked TC (or “In”) is calibrated to hold exactly its nominal volume. If you pour the liquid out into a second vessel, the volume delivered will be slightly less than 100 mL because a thin film of liquid remains adhered to the glass walls. For precise transfers, you should always use a pipette or burette marked TD (or “Ex”), which accounts for this residual drainage film in its calibration.


